AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Entropy definition biology10/13/2023  If the balls are arranged in a corner, the entropy will be low, as the degree of disorganization in this system is also low. The image of a box containing balls provides a good analogy for understanding the concept of entropy. The more disordered the system, the greater its entropy. 
It is what characterizes the degree of organization (or disorganization) of any physical system. 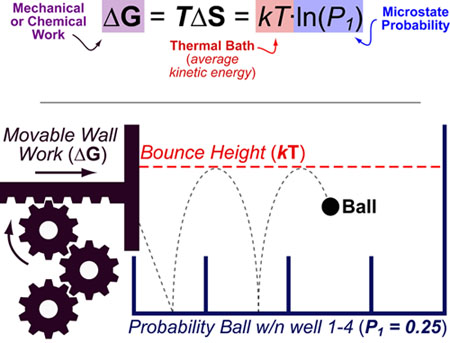
To find out how much of this energy can be considered ?free? available for consumption?, it is necessary to know another concept: that of entropy.Įntropy is related to order and disorder in a system. But not all the energy generated is available to be turned into useful work. For appliances to work, it depends on electricity. To move a car, for example, it is necessary to obtain energy by burning fuel. Proposed in the mid-1980s as a way to generalize usual statistical mechanics, a new formula successfully generalizes the application of entropy to phenomena as disparate as cyclones and giant molecules.Įnergy is one of the concepts in physics with the most visible application in everyday life. THE entropy, a concept as rich as it is mysterious, explains, for example, how the energy contained in a piece of coal can move a locomotive, or why, in order to cool the water, the refrigerator heats up on the outside. In the food coloring example given above, part of the food coloring solution may be evaporating, a process separate from the uniform distribution of a solute. Theoretically, this can be measured, but in practice it is very difficult to create an absolutely closed scenario. This must happen in a "closed" system where no energy enters or leaves. THE entropyassesses the amount of clutter, understood as a change in heat from an earlier point to a later point in time. In more technical terms, entropy is a specific value that measures how much energy is released into a system when it settles at the lowest potential energy. The object always ends up with the lowest potential energy position possible, such as resting on the ground. It is converted into kinetic energy of movement as the object falls. A high position has greater potential energy. 
Unless we put energy into a system, like an arm and a ball, holding an object, it falls towards the ground. When you drop food coloring into a glass of clear water, even if you don't stir it, this unified concentration of one drop gradually spreads out until each part of the water has the same color density.Īnother type of entropy that has to do with visible movement (as opposed to the invisible movement of heat) involves gravity. For example, molecules always spread out to uniformly fill a container. Change always involves moving from imbalance to balance, consistent with moving to descending order. Temperature is not the only transformation involved in entropy. This is due to entropy, as water molecules tend to use their accumulated potential energy, release heat and have a lower potential energy. If the heat source is removed, we can all guess that the water will gradually cool down to room temperature. You're adding heat, a form of kinetic energy, to accelerate molecules in water. For example, water will only boil as long as you hold a pot over the flames. Systems can only maintain organization on a molecular level while energy is added. This definition gives us an idea of why things seem to change for no reason. In physics, entropy is a mathematical measure of a change from the greatest to the smallest potential energy, related to the second law of thermodynamics.Įntropy comes from a Greek word meaning "transformation". 
THE entropy it can affect the space in which a substance spreads, its phase change from solid to liquid to gas, or its position. In your day-to-day, you intuitively understand how the entropy whenever you add sugar to coffee or melt an ice cube in a glass. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
